
The more salt there is dissolved in the water, the. Salinity describes how much salt is dissolved in a sample of water. This is represented by the addition of red spheres and blue cubes to the box from Fig. In addition, the molal ratio between water and ions is decreased, resulting in a low interaction of the water molecules with the electrode at the interface, thus inhibiting the water-splitting considerably. When salt is dissolved in fresh water, the density of the water increases because the mass of the water increases. Other factors affect waters density such as whether it is tap or fresh water or salt water. Water has the maximum density of 1 g/cm 3 only when it is pure water. When the temperature changes from either greater or less than 4 degrees, the density will become less then 1 g/cm 3. The formation of ionic aggregates owing to the increasing cohesive energy in WiSE disturbs the hydrogen-bond network resulting in physicochemical changes in the bulk liquid phase. Water has its maximum density of 1g/cm 3 at 4 degrees Celsius. Larger ESW values are possible due to the formation of a solvent blocking interface (SBI). A carbon-based symmetric supercapacitor filled with WiSE showed a larger electrochemical stability window (ESW), allowing to span the cell voltage and specific energy. Simulations covered the concentrations range of 1 mol dm −3 (1 mol kg −1) to 8 mol dm −3 (15 mol kg −1), demonstrating a significant increase in viscosity and density and reduction in ionic conductivity as the concentration reaches the WiSE conditions. 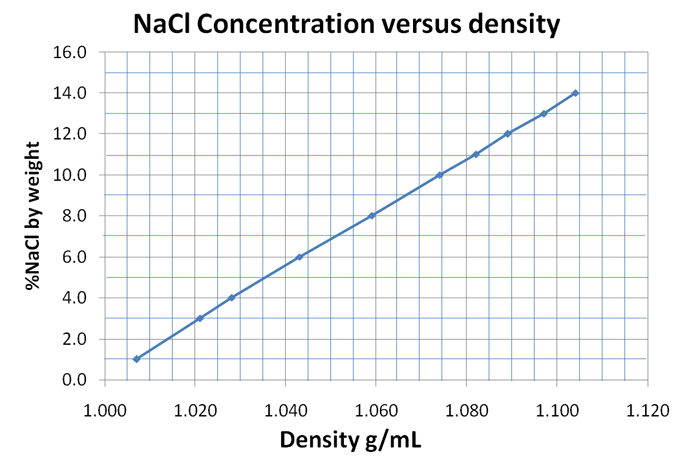 
With salinity, the density of saltwater changes as well. Seawater can reach a density of 1050 kg/m 3 or greater deep in the ocean, under extreme pressure. Atomistic molecular dynamics simulations were employed to investigate the energetic, structural, and transport properties of aqueous electrolytes based on sodium perchlorate (NaClO 4). The density of salt water is 1023.6 kg/m 3 at a temperature of 25 ☌, a salinity of 35 g/kg, and a pressure of 1 atm. We report here molecular dynamics simulations combined with in situ experimental studies to understand the advantages and disadvantages of replacing conventional (salt-in-water, SiWE) aqueous-based electrolytes with very concentrated (water-in-salt, WiSE) systems in supercapacitors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
